Qureight and Vicore Pharma release results characterising lung disease patients who may be more responsive to C21 treatment
Qureight and Vicore Pharma publish results indicating that patients with earlier stage idiopathic pulmonary fibrosis (IPF) are more likely to have improvement in lung function in response to treatment with Vicore’s novel angiotensin II type 2 agonist, C21.

- Qureight and Vicore Pharma publish results indicating that patients with earlier stage idiopathic pulmonary fibrosis (IPF) are more likely to have improvement in lung function in response to treatment with Vicore’s novel angiotensin II type 2 agonist, C21.
- C21 is an oral small molecule in development for treatment of IPF.
- Interim data from Vicore’s phase 2a AIR trial was analysed using Qureight’s proprietary platform and AI technology.
- The Qureight platform was used to analyse CT scans of AIR trial patients to quantify biomarkers associated with IPF progression.
- Combining analysis of CT scans and blood biomarkers with clinical trial data in this way provides unique insight into how IPF patients may respond to novel treatments.
At the 2023 ERS International Congress in Milan, Qureight and Vicore Pharma will release the results of interim data analysis from the phase 2a AIR trial with C21 for idiopathic pulmonary fibrosis (IPF).
The aim of this analysis was to find signals in historic CT scans that could identify IPF patients more likely to respond to Vicore’s novel angiotensin II type 2 agonist, C21, an oral small molecule in development for treatment of the disease.
Qureight’s proprietary data platform and AI technology was used to analyse historic CT scans of the AIR trial patients to quantify biomarkers associated with IPF progression.
The results suggested that patients with early IPF, and thus more limited disease, are more likely to have improvement of lung function following treatment withC21.
About AIR
AIR is Vicore Pharma’s ongoing phase 2a, multi-center, open-label, single-arm trial to evaluate the safety and efficacy of novel angiotensin II type 2 receptor agonist C21 for treatment of patients with IPF.
Treatment is over 24 weeks, with an optional extension to 36 weeks. Efficacy is assessed using forced vital capacity (FVC) measurements, and results are compared to average untreated FVC decline of 60ml per 12 weeks.
Method of Analysis
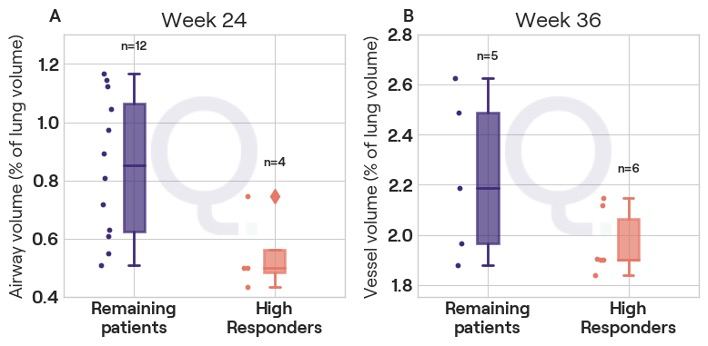
The Qureight platform enables automated segmentation of CT scans for volumetric quantification of lung, airway, vessel, and fibrosis volumes in the lungs of IPF patients.
The analysis of interim AIR results identified a group of patients with improved lung function following 24 weeks of treatment with C21. Automated segmentation of historic CT scans of these ‘high responder’ patients was performed – using the Qureight platform – to quantify the volumes lungs, airways, blood vessels, and fibrotic tissue.
Results
The results showed that the high responder group had lower airway volumes at baseline when compared to the remaining patients.
These results suggest that patients with early IPF, and thus more limited disease, may respond better to C21. These findings are consistent with C21’s mechanism of action - stimulation of the AT2 receptor on alveolar epithelial type 2 cells, inducing a reparatory mechanism with subsequent improvement in FVC
Dr. Muhunthan Thillai, co-founder and CEO of Qureight, said:
“These results show how the Qureight platform and our AI can be used to enhance the data available from a clinical trial.Qureight’s technology enables pharmaceutical companies to identify the ‘hidden patterns’ in their data, which in turn helps to accelerate the drug development process and ensure that IPF patients receive better, more effective treatments.”
Dr. Rohit Batta, CMO of Vicore Pharma, said:
“We are very pleased to have worked with Qureight to generate these valuable insights around the link between disease progression and therapeutic efficacy of C21. These insights will serve us well as we continue our work to advance C21 for the treatment of IPF into our Phase2b ASPIRE study.”
For more information on these results, or to learn more about our work with Vicore Pharma, contact a member of the Qureight team.
